Science is the poetry of reality. There are lots and lots of phenomenons happening all around you at the very moment you are reading this article. Science has explanations for everything possible. One such mind-blowing concept is of Magic Numbers.
Let’s discuss in detail about the concept of Magic Number.
Introduction

The idea of Magic numbers in the field of science alludes to a particular property, (for example, dependability) for just certain agents among a conveyance of structures. It was first perceived by examining the power of mass-spectrometric signs of uncommon gas bunch particles.
On the off chance that a gas gathers into groups of particles, the quantity of molecules in these bunches that are well on the way to frame fluctuates between a couple and hundreds.
Be that as it may, there are tops at explicit bunch sizes, veering off from an unadulterated measurable conveyance. In this manner, it was reasoned that bunches of these particular quantities of uncommon gas molecules command because of their excellent soundness.
The idea was likewise effectively applied to clarify the monodispersed event of thiolate-secured gold bunches; here the extraordinary solidness of explicit group sizes is associated with their separate electronic design.
The term magic numbers are likewise utilized in the field of atomic physics. In this specific circumstance, magic numbers frequently speak to three-dimensional figurate numbers, for example, the octahedral numbers: they include the quantities of circles in circle packings of Platonic solids and related polyhedra.
In atomic physics, a magic number is various nucleons (either protons or neutrons, independently) to such an extent that they are masterminded into complete shells inside the nuclear nucleus.
Subsequently, a nuclear nucleus with a ‘magic ‘ number of protons or neutrons is significantly more steady than other nuclei. The seven most broadly perceived magic numbers starting in 2019 are 2, 8, 20, 28, 50, 82, and 126.
For protons, this relates to the components helium, oxygen, calcium, nickel, tin, lead, and the theoretical unbihexium, albeit 126 is so far just known to be a magic number for neutrons.
Nuclear nucleus comprising of such a magic number of nucleons have a higher normal restricting vitality per nucleon than one would expect dependent on forecasts, for example, the semi-observational mass recipe and are henceforth more steady against atomic rot.
The abnormal dependability of isotopes having magic numbers implies that transuranium components could hypothetically be made with very enormous nucleus but then not be dependent upon the incredibly quick radioactive rot regularly connected with high nuclear numbers. Enormous isotopes with magic quantities of nucleons are said to exist in an island of strength
History
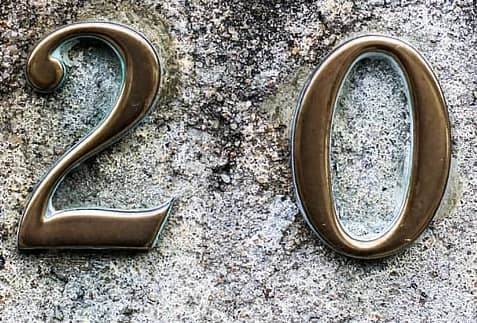
After dealing with the Manhattan Project, the German physicist Maria Goeppert Mayer got intrigued by the properties of atomic splitting items, for example, rot energies and half-lives. In 1948, she distributed a collection of test proof for the event of shut atomic shells for nucleus with 50 or 82 protons or 50, 82, and 126 neutrons.
Two years after the fact, in 1950, another distribution followed in which she ascribed the shell terminations at the magic numbers to turn circle coupling.
As per Steven Moszkowski (an understudy of Maria Goeppert Mayer), the expression “magic number” was instituted by Wigner: “Wigner also put stock in the fluid drop model, yet he perceived, from crafted by Maria Mayer, the exceptionally solid proof for the shut shells.
It appeared to be similar to magic to him, and that is the manner by which the words ‘Magic Numbers’ were coined.”
These magic numbers were the bedrock of the atomic shell model, which Mayer created in the next years along with Hans Jensen and finished in their mutual 1963 Nobel Prize in Physics.
Doubly Magic
Nucleus which have neutron number and proton (nuclear) numbers each equivalent to one of the magic numbers are designated “doubly magic”, and are particularly steady against decay. The realized doubly magic isotopes are helium-4, helium-10, oxygen-16, calcium-40, calcium-48, nickel-48, nickel-56, nickel-78, tin-100, tin-132 and lead-208.
In any case, just the principal, third, fourth, and last of these doubly magic nuclides are totally steady, in spite of the fact that calcium-48 is incredibly extensive and in this manner normally happening, deteriorating just by a wasteful twofold beta less rot process.
Doubly-magic impacts may permit the presence of stable isotopes which in any case would not have been normal. A model is calcium-40, with 20 neutrons and 20 protons, which is the heaviest stable isotope made of a similar number of protons and neutrons.

Both calcium-48 and nickel-48 are doubly magic since calcium-48 has 20 protons and 28 neutrons while nickel-48 has 28 protons and 20 neutrons. Calcium-48 is very neutron-rich for such a light component, however, like calcium-40, it is settled by being doubly magic.
Magic number shell impacts are found in conventional plenitudes of components: helium-4 is among the most plentiful (and stable) nucleus in the universe and lead-208 is the heaviest stable nuclide.
Magic impacts can keep precarious nuclides from rotting as quickly as would some way or another be normal. For instance, the nuclides tin-100 and tin-132 are instances of doubly Magic isotopes of tin that are shaky and speak to endpoints past which soundness drops off quickly.
Nickel-48, found in 1999, is the most proton-rich nuclide known past helium-3. At the other extraordinary, nickel-78 is additionally doubly magic, with 28 protons and 50 neutrons, a proportion watched distinctly in a lot heavier components separated from tritium with one proton and two neutrons (78Ni: 28/50 = 0.56; 238U: 92/146 = 0.63).
In December 2006, hassium-270, with 108 protons and 162 neutrons, was found by a global group of researchers drove by the Technical University of Munich, having a half-existence of 9 seconds.
Hassium-270 clearly frames some portion of an island of soundness, and may even be doubly magic due to the distorted (American football-or rugby ball-like) state of this nucleus.
In spite of the fact that Z = 92 and N = 164 are not magic numbers, the unfamiliar neutron-rich nucleus uranium-256 might be doubly magic and round because of the distinction in size among low-and high-rakish force orbitals, which modifies the state of the atomic potential.
Science has its own infinite possible ways to amaze every human mind in this universe. Just opening your mind a little towards science will open a path to an unbelievable, yet a believable world of magic and great discoveries.
Also read :
Why Stars Twinkle But Planets Don’t? – Exploring The Infinite Universe
Factors Affecting Friction – A Necessary Evil
Very informative
this post is very useful to us thanks for sharing this info with us…